The Clinical screen contains drug manufacturer information, FDA Drug Application Type, generic and Tall Man descriptions, drug notes, customized warning labels, monographs and other clinical information.
To add/view Clinical Information:
1. Click ![]() on the Drug Record
vertical icon bar.
on the Drug Record
vertical icon bar.
2. The Clinical screen displays:
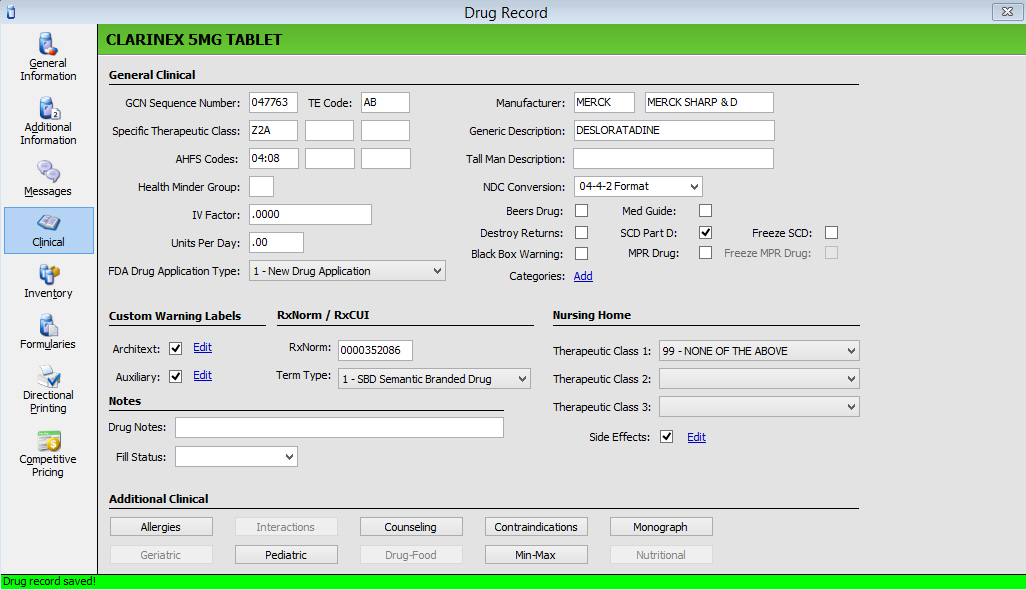
3. Use the following definitions to add or edit information in each field as needed:
| DRUG RECORD/CLINICAL SCREEN | |
General Clinical |
|
| Field Name | Field Definition |
| GCN Sequence Number | System generated entry displays GCN Sequence Number for the drug. GCN Sequence Number indicates same active ingredients, route of administration, drug form and strength. Numbers can be updated manually. |
| TE Code | Four-character, editable field only populated if authorized for clinical updates. The field is populated with the Orange Book code during the Service Pack 25 update. |
| Specific Therapeutic Class | Type the drug therapeutic class code. This code checks for duplicate therapy during prescription processing. |
| Health Minder Group (HMG) | Type special group codes to apply to selected maintenance medications if you are using the Health Minder Form to remind patients about refills on maintenance medications. |
| IV Factor | Type the factor used to convert the number of milliequivalents ordered for the drug into the number of milliters needed. |
| Units Per Day | type the number of capsules, tablets, milliters, etc. which constitute a normal daily dose. An overdose warning displays when this amount is exceeded by the quantity per day assigned to a SIG. |
| FDA Drug Application type | First
Databank maintained field. This field indicates whether a drug
was approved under a New Drug Application (NDA) or an Abbreviated
New Drug Application (ANDA). Select entry from the drop-down list:
1 = NDA 2 = ANDA 3 = Other 4 = Not Defined |
| Manufacturer | Type the manufacturer's code for the drug. The manufacturer displays in the second position. To print the manufacturer, a manufacturer's code routine must be in the label routine. |
| Generic Description | Type the generic drug description. |
| Tall Man Description | Type the Tall Man drug name. |
| NDC Conversion | Code used to add a zero to barcoded NDCs containing only 10 digits. Select Format from the drop-down list. |
| Beers Drug | Indicates that the drug is considered to be potentially inappropriate to use in older adults. During Clinical Checking, the pharmacist is alerted if filling for a Beers Drug for a patient over 65 years old. |
| Med Guide | Check if this drug requires a MedGuide. |
| Destroy Returns | Check this box if this drug is identified as a drug that cannot be returned to inventory and should be destroyed if returned. |
| SCD Part D | Check
this box if this drug is identified as a Short Cycle Dispensing
(SCD) Part D drug. This field is updated by the following programs:
|
| MPR Drug | If a drug is added to the system and has one of the specified drug classes, the field is automatically selected. Clinical updates apply. |
| Freeze SCD | Check this box to freeze the SCD Part D field when processing any system updates. |
| Freeze MPR Drug | Check this box to stop the system from applying Clinical Updates to drugs with the MPR field selected. |
| Black Box Warning | Check
this box if this drug has an associated FDA mandated warning that
appears on the package insert indicating the drug may have serious,
or even life-threatening, side effects. Added the black box warning
when updating/adding a drug via the internet and clinical data
updates.
The first way to determine if a drug has an associated black box warning is to check whether the drug's patient education monograph has a 'W' section. The 'W' section of a monograph contains the text of the black box warning itself. The second way to determine if a drug has a black box warning is to determine whether the drug's prioritized label warning includes a label warning with the code 0298. This label warning has the text 'Read the FDA black box warning information for the medication.' |
| Categories | Click Add to create drug categories used to group drugs together. To group drugs, the RPh or Technician must have Drug Access/Category Update checked in Security Access. |
| Customized Warning Labels | |
| Architext | A check displays in the field if the drug has Architext Label Warnings attached. Click Edit to display Customized Warnings and Prioritized Warnings. The Prioritized Warnings are the label codes from First Databank and are listed in the priority assigned by First Databank. A single drug may have up to 10 prioritized warnings. The Customized Warnings field is used if you want the codes in a different order than that from First Databank. Up to five Customized Warnings can be saved for each drug. To move a warning from the Prioritized column, select the warning and click Add. The warning now displays in the customized column. Warnings print in this order and will not change if you subscribe to Clinical Updates. |
| Auxillary | A check displays in the field if the drug has Intercon Label Warnings attached. Click Edit to display Customized Warnings and Prioritized Warnings. The Prioritized Warnings are the label codes from First Databank and are listed in the priority assigned by First Databank. The Customized Warnings field is used if you want the codes in a different order than that from First Databank. To move a warning from the Prioritized column, select the warning and click Add. The warning now displays in the customized column. Warnings print in this order and will not change if you subscribe to Clinical Updates. |
| Nursing Home | |
| Therapeutic Class 1 | Nursing Home Therapeutic Class Codes are used to categorize drugs according to Federal Government guidelines. Field is used in conjunction with the Nursing Home Consultant Worksheet. Up to three classes for each drug can be entered. Select code from the drop-down list. |
| Therapeutic Class 2 | Same as above. |
| Therapeutic Class 3 | Same as above. |
| Side Effects | Field is used to prioritize side effects. Click Edit to view Drug Side Effects. To add a side effect to the drug, select the drug in the Side Effects Scan and click Add to Selection. To prioritize the selected side effects, select the effect and click Move Up or Move Down to place in the correct order. |
Notes |
|
| Drug Notes | Fifty-character alphanumeric field for user-defined notes. |
| Fill Status | Select the status from the drop-down list. This status is added to the Drug Notes. |
| Additional Clinical | The clinical fields are drug-specific and are updated via Clinical Updates. |
| Allergies | |
| Interactions | |
| Counseling | |
| Contraindications | |
| Monograph | |
| Geriatric | |
| Pediatric | |
| Drug-Food | |
| Min-Max | |
| Nutritional | Nutritional Clinical Warnings display if you have a subscription to NutriLinkTM. |
 Architext
Warning Labels and Descriptions
Architext
Warning Labels and Descriptions
Drug Specific Therapeutic Class Codes and Descriptions
American Hospital Formulary Service Codes (AHFS). QS/1 updates this information from FDB but it does not display in the system. However, AHFS Codes are available as a Report Select Option in the Drug File.
4. Press CTRL+S or click ![]() .
.
Return to Create Drug Records in PrimeCare